CYTOBALANCE by Bioscience Institute detects cytokines increase in healthy people, that is without chronic diseases symptoms nor cancer prodromic genetic alterations. With CYTOBALANCE it is possible to identify or prevent physiological or pre-pathological conditions predisposing to the development of such symptoms or genetic alterations.
LOW-GRADE CHRONIC INFLAMMATION
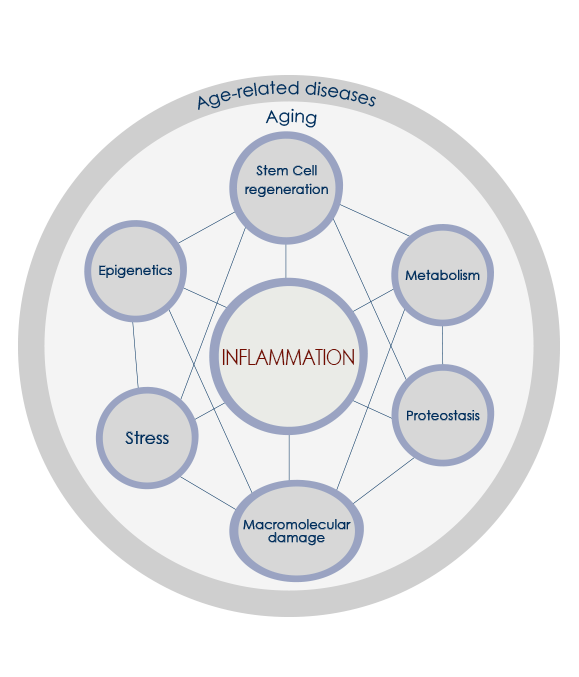
Inflammation is not just a damage or infection response, and it can be stimulated by triggers other than viral and bacterial antigens. With life expectancy increasing, immune system must counteract all these triggers for a long time; this could make inflammation increases, giving rise to the so-called “low-grade chronic inflammation”. Low-grade chronic inflammation is among the most important causes of frailty, diseases and age-related deaths. Among diseases associated with this type of inflammation are atherosclerosis, Alzheimer’s and Parkinson’s disease, type 2 diabetes, and cancer. By counteracting it, it is possible to live longer and with the highest attainable standard of health.

L’Inflammaging
Age-related low-grade chronic inflammation that is not associated with infections is called inflammaging. It is mostly triggered by endogenous signals chronically activating immune system, such as cellular debris or oxidized proteins.
Nutrient excess and overnutrition can contribute to this phenomenon. They trigger a specific kind of chronic inflammation (the so-called “metabolic inflammation”, or “metaflammation”) that is associated with metabolic diseases such as obesity and type 2 diabetes.
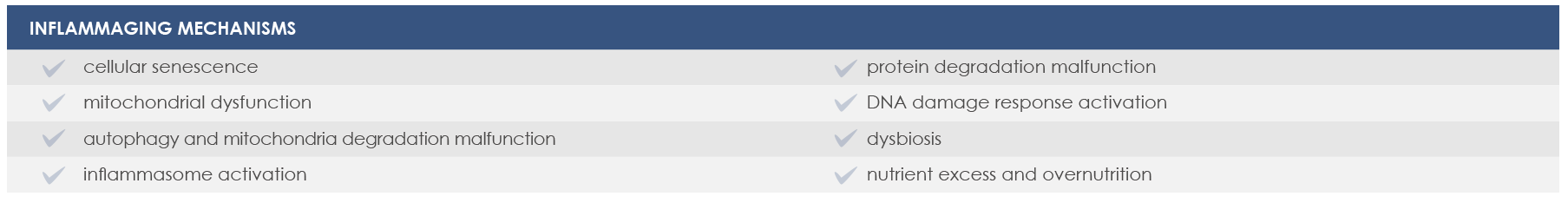
All inflammaging-promoting stimuli converge on a limited number of sensors, which in turn trigger inflammation and metabolic responses via innate immunity mechanisms. Until middle age organism survival strictly depends on this process. However, in post-reproductive age inflammatory response increase often becomes detrimental. The increase and accumulation of senescent cells play a role too. Moreover, during aging the ability to resolve inflammation decreases. Finally, according to the garbaging theory, the age-related progressive impairment of cell debris elimination systems largely sustains inflammaging.
Inflammation and metabolic Endotoxemia
Chronic inflammation progressively increases during years or decades. Meanwhile, inflammaging is promoted by nutrient excess and overnutrition, and metabolic inflammation contributes to insulin resistance and activates inflammatory responses in organs and tissues such as the brain, the pancreas, the liver, muscles and adipose tissue.
Dietary fats play a pivotal role. They are associated with the increase of oxidative stress, cytokines and lipopolysaccharide – LPS, the endotoxin associated with sepsis and metabolic endotoxemia, a condition associated with low-grade inflammation and cardiometabolic issues. Also irregular meals can contribute to metabolism and inflammatory response changes. Finally, a nutrient-dense diet promotes adipocyte size increase until a critical point where these cells contribute to metabolic inflammation too.
Low-grade inflammation-associated reduction of immune functions and proinflammatory cytokines increase negatively impact metabolism, bone density, strength, exercise tolerance, vascular system, cognitive functioning, and mood. That is why among inflammaging-related diseases are diabetes, cardiovascular diseases, osteoporosis, and dementia.

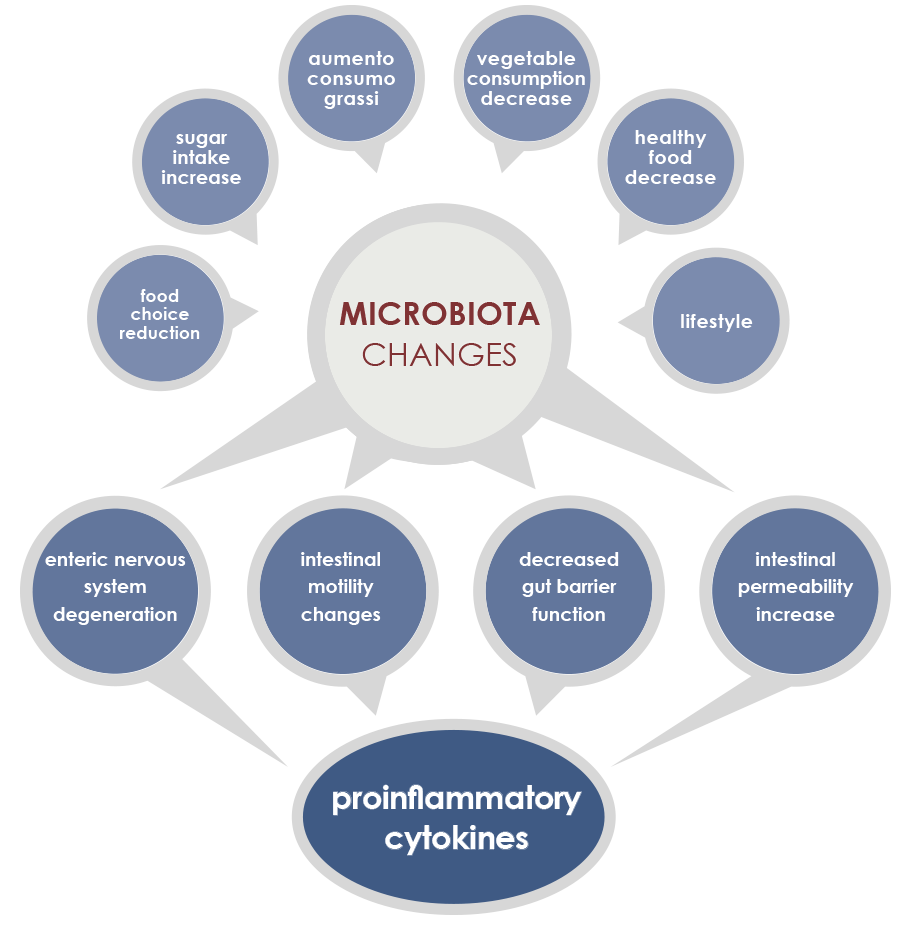
Gut microbiota and inflammaging
Gut microbiota is involved in both induction and maintenance of inflammaging. The mechanisms underlying this relationship include phenomena associated to the majority of age-associated diseases (enteric nervous system degeneration, gut motility alterations, decreased efficiency of the mucosal barrier) that are linked to increased proinflammatory cytokines and to changes in gut microbiota composition and stability.
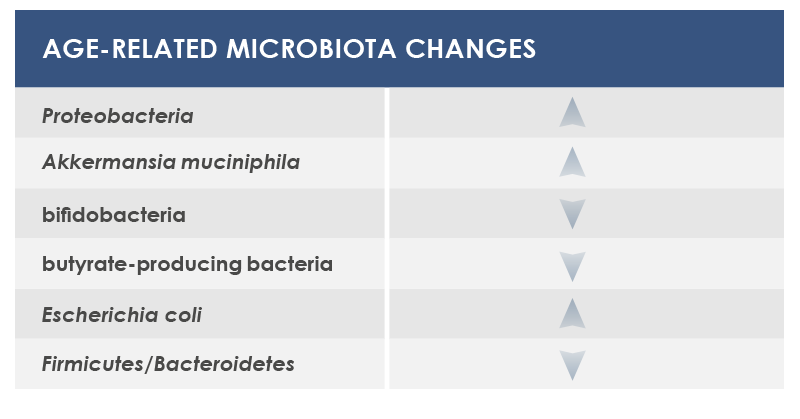
In particular, during aging microbiota tends to lose the ability to ferment carbohydrates, to better ferment proteins, and to have a reduced diversity, with the increase of some bacteria species. External factors exert a little influence on some microbiota changes; that is why these shifts could be considered core features of elderly microbiota. Other changes are instead more dependent on lifestyle factors, for example on eating habits and drugs.
Among bacteria whose levels could change are included: Proteobacteria, that are associated to both local and systemic inflammation; Akkermansia muciniphila, that breaks down a gut barrier component, mucin; bifidobacteria, health-friendly lactic ferments; microbes producing butyrate, an inflammation regulator; and Escherichia coli.
In the blood, molecules from gut microbiota activate macrophages, generating a proinflammatory status that promotes atherosclerosis and that is associated with both cardiovascular diseases and vascular dementia. Moreover, cytokines and short-chain fatty acids variations associated with gut microbiota changes influence cognitive decline. Finally, the proinflammatory response stimulated by lipopolysaccharide promotes beta-amyloid protein production; this is an evidence of a possible link between gut microbiota and Alzheimer’s disease.
inflammation and mitochondrial DNA
Also mitochondrial DNA (mtDNA) can induce inflammation. It can exit from mitochondria because of cellular stress or necrosis, but evidence suggests phenomena such as programmed necrosis and mitochondrial release from platelets could be involved too. Moreover, inflammaging is promoted by aging; in fact released mtDNA level gradually increases after age 50.
mtDNA and bacterial DNA are similar; that is one reason why mtDNA promotes inflammation. Moreover, also mtDNA oxidization stimulates immunity; as a consequence, oxidized mtDNA promotes inflammation.
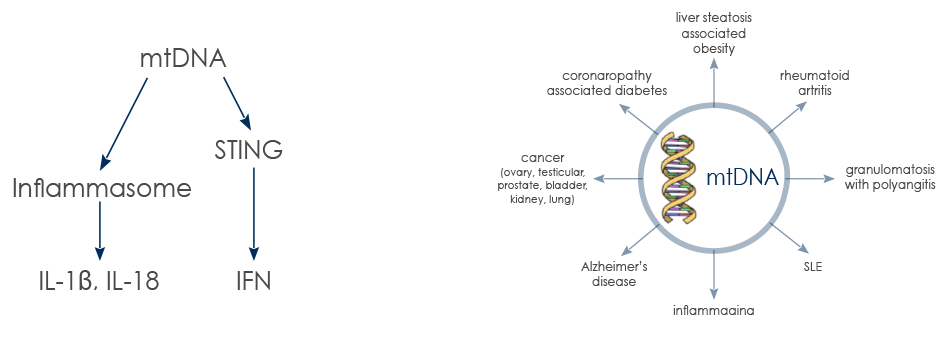
Proinflammatory action of mtDNA is mediated by Toll-Like Receptor 9 (TLR-9) and can involve Stimulator of INterferon Genes (STING) pathway.
Elevated mtDNA levels in the blood are associated with systemic and organ-specific chronic diseases. mtDNA inflammatory action can be boosted by the highly oxidant extracellular environment typical of chronic diseases.

chronic inflammation and cancer
More than 20% of all cancers is initiated or exacerbated by inflammation: stomach cancer is associated with gastritis, colorectal cancer with colitis, hepatocellular carcinoma with chronic inflammation, and almost 20% of smokers with bronchitis can develop cancer. Conversely, NSAIDs are associated with a 30-60% reduction in colorectal cancer incidence.
Inflammation acts both via extrinsic pathways (that is, via chronic conditions associated with smoldering, non-resolving inflammation) and intrinsic pathways (that is, genetic events that orchestrate the generation of cancer-related inflammation).
Moreover, chronic inflammation is associated with immunosuppression, a known cancer risk factor. Its effect is mediated by reactive oxygen and nitrogen species (ROS and RSN), by DNA methylation changes, and by a range of molecules produced by immune cells. In particular, ROS and RSN damage DNA, proteins and lipids; they act also on stem cells, and in this way they promote the development of aggressive cancer and a vicious circle of inflammation and oxidative stress playing a pivotal role in cancer development. Moreover, ROS and RSN can promote a generalized DNA demethylation, which is linked to another cancer risk factor: genome instability. At the same time, proinflammatory cytokines can reduce the expression of genes and microRNA involved in tumor suppression, making methylation increases.
All these phenomena lead to cancer cell growth and survival, angiogenesis, tissue invasion, and metastases, or to anticancer immune response suppression.
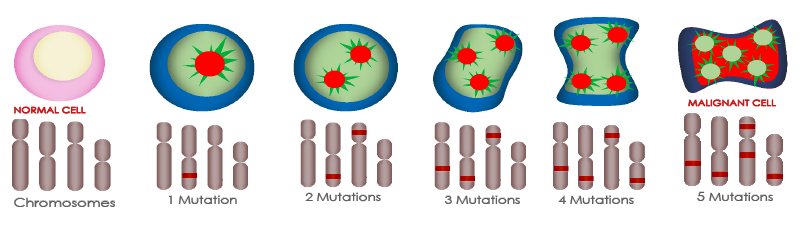
Progressive mutations in cancer cell development
Inflammation biomarkers and cancer
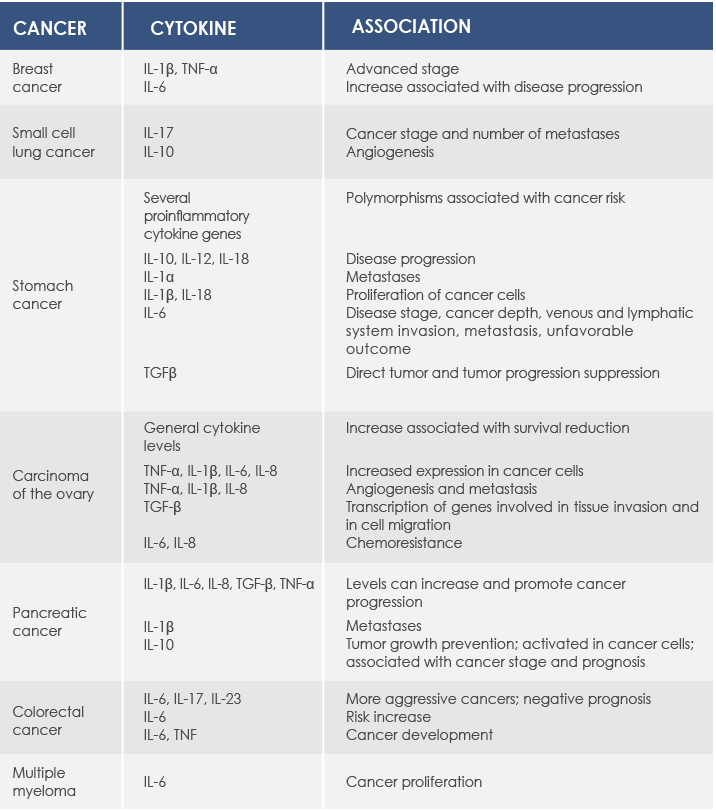
Several molecular biomarkers of inflammation are associated with cancer. In particular, proinflammatory cytokines are produced by both cancer cells and cells in cancer microenvironment such as Tumor Necrosis Factor α (TNF-α)-producing tumor-associated macrophages. TNF-α is a cytokine produced in the first phase of inflammatory response playing a pivotal role in maintaining chronic inflammation; moreover, it is associated with cancer growth, angiogenesis, tissue remodeling, and metastases. Proinflammatory cytokines regulate C reactive protein (CRP) production too.

In healthy people CRP levels are very low, but they can rapidly increase with inflammation.
They are associated with cancer progression and survival reduction in several types of cancer (esophagus, stomach, colorectal, liver, pancreas, and ovarian). Higher CRP is associated with a greater risk of cancer development (for example, colorectal or breast cancer) and with colorectal cancer mortality.
Come riconoscere l'infiammazione cronica
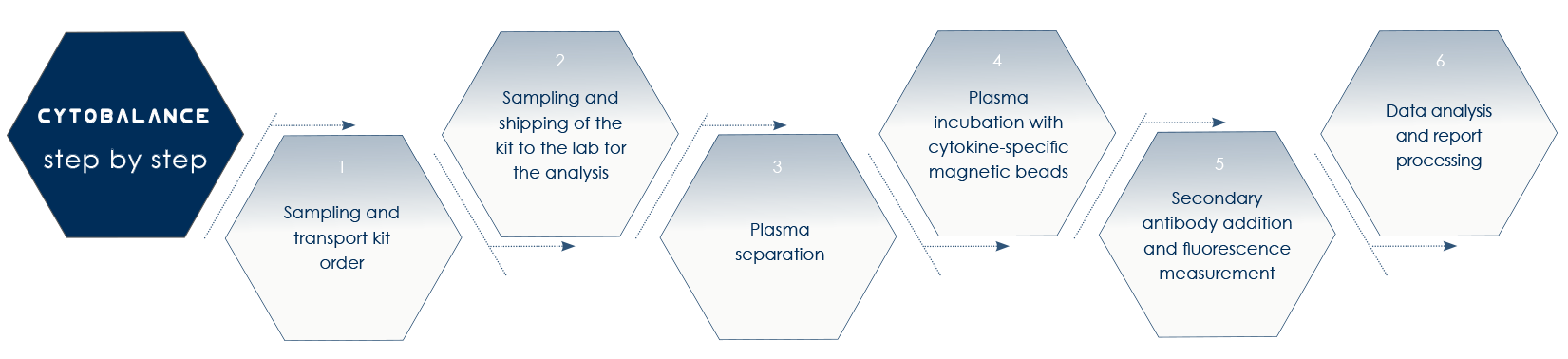
L’infiammazione cronica di basso grado non è associata dai classici sintomi dell’infiammazione acuta (gonfiore, dolore, arrossamento e calore), ma numerosi marcatori biomolecolari (tra cui diverse citochine e la PCR) permettono di riconoscere la sua presenza grazie ad una semplice analisi del sangue. Con CYTOBALANCE Bioscience Institute propone il monitoraggio di 13 diversi marcatori dell’infiammazione.
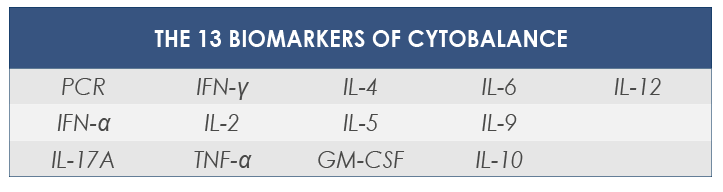
GM-CSF - Granulocyte-Macrophage Colony-Stimulating Factor promotes the growth of several precursors of blood cells, for example granulocytes and macrophages ones.
TNF-α - Low TNF-α levels are associated with the increase of adhesion molecules on the surface of endothelial cells; this makes leucocytes move from blood to the infection site. Moreover, TNF-α activates these leucocytes and stimulates the production of lymphocyte-recruiting cytokines by mononuclear phagocytes. It is involved in cancer immunosuppression and promotes cancer growth and epithelial-mesenchymal transition.
IL-2 - Interleukin-2 is the principal mediator of G1 to S cell cycle phase transition in T cells. It is an autocrine T cell growth factor, stimulates the production of other cytokines, promotes NK cells growth, and increases their cytotoxic activity.
IL-4 - Interleukin-4 regulates immune response by acting on several types of cells, such as T and B cells, macrophages, and endothelial cells. Among its actions are Th2 cells growth and differentiation, class II MHC antigens expression increase on B cells, and inflammation cells recruiting via regulation of the expression of adhesion molecules (V-CAM-1) on endothelial cells.
IL-5 - Interleukin-5 induces eosinophils differentiation and IgA production increase. It is involved in the stimulation of B cells growth.
IL-6 - Interleukin-6 is the most important activator of lymphocyte differentiation in plasmocytes and IgA-producing cells. It induces hemopoietic, megakaryopoietic, and myelopoietic progenitors cell cycles, and stimulate acute phase proteins production in liver cells. High IL-6 levels can increase the susceptibility to cancer (for example hepatocellular carcinoma, multiple myeloma, and lung cancer).
IL-9 - Interleukin-9 induces T helper cells development.
IL-10 - Interleukin-10 limits and can terminate inflammatory reactions, promoting
pathogens eliminations and limiting infection-associated damage. It can exert different effects, depending on its expression duration, level, and localization: sometimes it acts as immunosuppressor, whereas in other cases it amplifies immune response and inflammationIL-12 - Interleukin-12 connects immune and adaptive immunity. It is the most important mediator of the early phase of innate and cell-mediated responses to intracellular microorganisms.
IL-17A - Interleukin-17A induces Granulocyte Colony-Stimulating Factor (G-CSF) and chemokine production. It plays an important role in chronic inflammation associated with autoimmune disease and allergy pathogenesis, and it is thought to have a pivotal role in psoriasis development. Scientific studies recently demonstrated that IL-17A increases in cancer patient serum; that is why it is supposed to have a significative prognostic value.
INF-α - Interferon α is involved in innate immune response against viral pathogens. Its presence is in part responsible for symptoms such as fever and muscle weakness.
IFN-γ - Interferon γ is the principal macrophage-activating cytokine. It is important both for innate immunity and for cell-mediated responses against intracellular microorganisms. It activates immune response effector cells and stimulates macrophage-mediates microorganisms killing.
PCR - In the presence of inflammation C reactive protein levels increase up to 1,000 fold. In particular, in case of severe tissue damage (such as cancer progression-induced one) CRP levels increase form 1 μg/ml to more than 500 μg/ml in 24-72 hours. It plays an active role in inflammation, and its levels have been using since long time as a marker of infections and of cardiovascular events in asymptomatic people. Moreover, CRP was associated with atherosclerosis, congestive heart failure, atrial fibrillation, myocarditis, aortic valve issues, and heart transplantation prognosis. Finally, its expression is elevated in the presence of other inflammation-associated diseases, such as rheumatoid arthritis.